Sketch of the particle–particle interaction for polar and apolar SiO 2... | Download Scientific Diagram
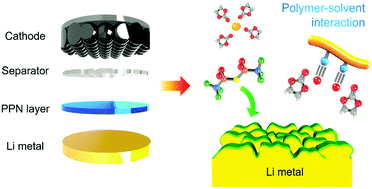
Polar polymer–solvent interaction derived favorable interphase for stable lithium metal batteries - Energy & Environmental Science (RSC Publishing)

Host-Enhanced Phenyl-Perfluorophenyl Polar−π Interactions | Journal of the American Chemical Society
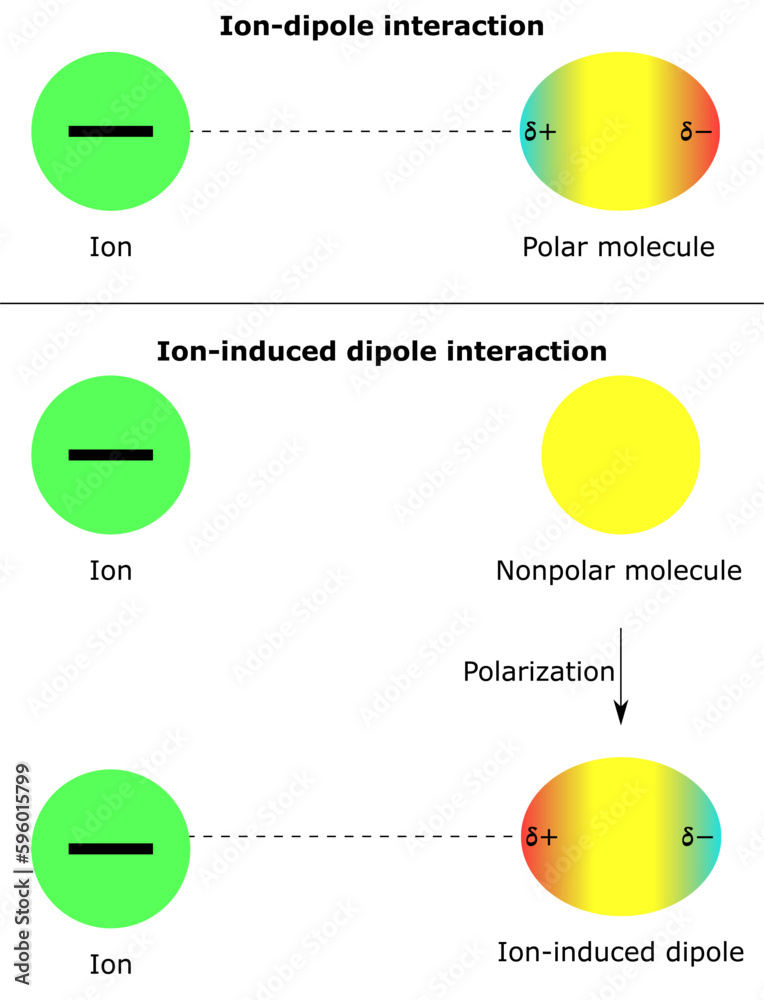
intermolecular forces ion dipole interaction ion induced nonpolar molecules polar partial positive negative charge physical chemistry physics Stock Vector | Adobe Stock

Host-Enhanced Phenyl-Perfluorophenyl Polar−π Interactions | Journal of the American Chemical Society
58.There is a line given that : Besides dipole dipole interactions, polar molecules can also interact by London forces. Thus the total intermolecular force in polar molecules increases. But How? London forces





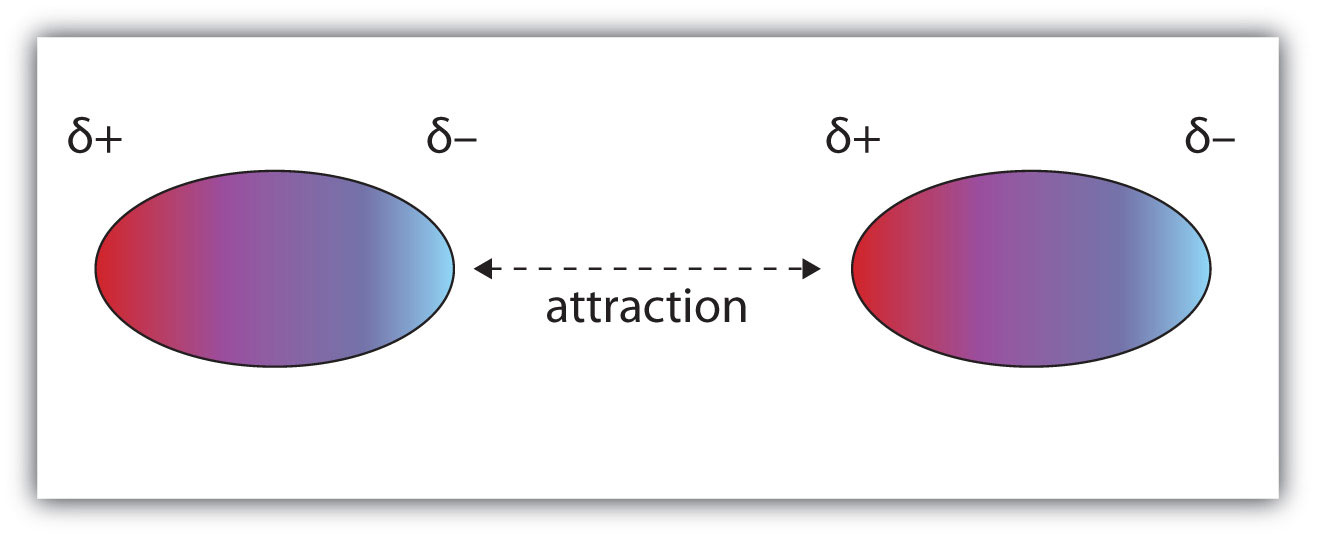


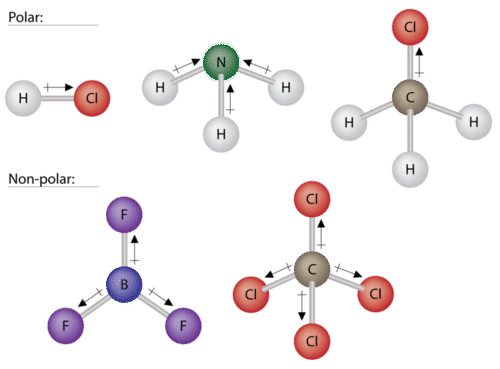
:max_bytes(150000):strip_icc()/PolarConvalentBond-58a715be3df78c345b77b57d.jpg)